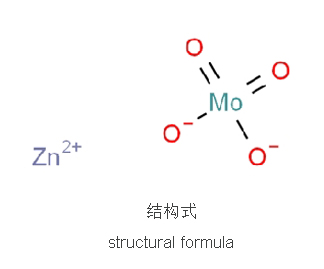
Zinc Molybdate

Zinc molybdate is an inorganic compound with molecular formula MoO4Zn and CAS number 13767-32-3. It is a white powder, and is hardly soluble in water, easily soluble in acid, and can undergo molybdate exchange reaction with metal oxides, such as reacting with cadmium oxide and magnesium oxide to produce cadmium molybdate and magnesium molybdate.
Molybdenum zinc oxide is a new type of anti-rust material with good tinctorial strength and hiding power. It is not only used as a primer and finishing coat, but also used molybdenum flame retardants and anticorrosion agent, startingmaterial for growing single crystals.
Basic Information

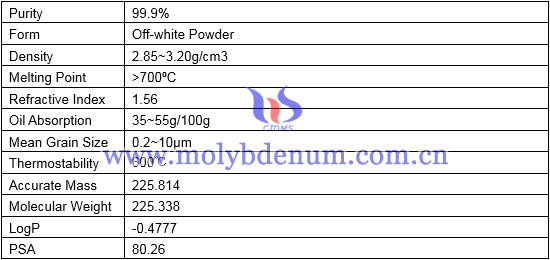
Physical & Chemical Properties
Health & Safety Information

Preparation
1.Synthesis method: zinc oxide reacts with sodium molybdate or molybdic acid, adding with a certain amount of body pigment at the same time. The finished product of zinc molybdate is obtained after washing, filtering, drying and crushing.
2.Zinc oxide and MoO3 react to form ZnMoO4 at 400℃, and then quickly obtain ZnMoO4 at 600~700℃.
Storage
Stored in normal temperature, sealed, cool, ventilated and dry.
Applications
1. Used in various occasions as corrosion inhibitor, anti-rust pigment, flame retardant and smoke suppressant, etc.;
2. Flame retardant and smoke suppressant for high molecular polymer and metal antirust white pigment;
3. Reagents used to manufacture alkaloids and other substances;
4. Used in dyes, molybdenum red pigments, catalysts, molybdenum salts and sun-resistant lake precipitation agents;
5. Used in the manufacture of flame retardants and metal corrosion inhibitors for pollution-free cooling water systems;
6. As an essential trace component for animals and plants.